Novavax Covid vaccine approved by FDA with new conditions
FDA places additional conditions on individuals who would be able to receive vaccine
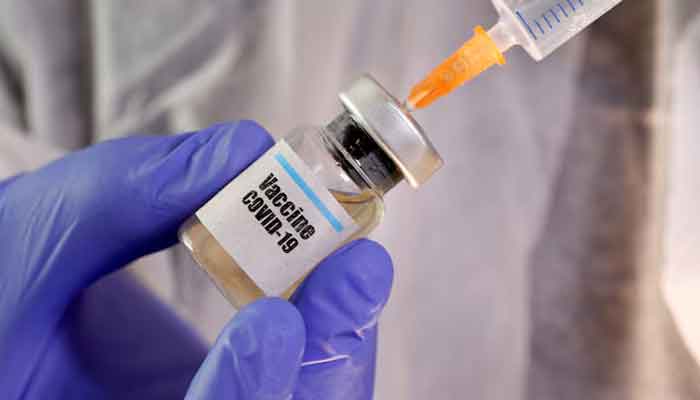
The US Food and Drug Administration approved Novavax's Covid-19 vaccine on Friday, but placed additional conditions on individuals who would be able to receive the vaccine.
The license restricts the use of the vaccine called Nuvaxovid to individuals aged 65 and older, and those between 12 and 64 who have at least one underlying condition that increases their risk of developing severe illness from Covid, according to the approval letter.
The letter did not specify what qualified as an underlying condition.
The FDA also deferred submission of pediatric studies from birth to less than 12 years for the application, as pediatric studies had not been completed.
Novavax CEO John Jacobs said the approval was a "significant milestone" that solidifies a path for people to access the vaccine.
The vaccine's prospects were thrown into doubt after the FDA missed its April 1 target to approve the shot. US Health and Human Services Secretary Robert F Kennedy Jr attributed the delay to the shot's composition in a CBS interview earlier that month.
Novavax, whose protein-based shot uses an older technology, missed out on the pandemic vaccine windfall – enjoyed by rivals Moderna and Pfizer which make messenger RNA-based vaccines – due to manufacturing issues and regulatory hurdles.
-
Meningitis leaves one dead, others critically ill; Know how it spreads
-
PCOS renamed PMOS: What new diagnosis means for millions of women’s health
-
Endometriosis linked to small increase in birth defect risk in Canadian study
-
Health Canada issues safety warning over baby self-feeding products sold on Amazon
-
Do psychopaths and others have same brain structure? Scientists reveal shocking details
-
Cruise ship Hantavirus outbreak leaves 3 dead, more infected
-
43 hair products tested: Researchers find cancer-linked chemicals in braiding hair
-
Study reveals how brain tells you to stop scratching
-
Hantavirus outbreak: MV Hondius reaches Tenerife for ‘unprecedented’ evacuation
-
Omega-3 supplements linked to faster cognitive decline in high-risk patients
-
Is the US at risk? Everything you need to know about the new Hantavirus quarantines
-
Breakthrough research suggests, gum disease can be prevented without killing good bacteria