This new medicine can treat types of pancreatic cancer
Pancreatic cancer symptoms include loss of appetite, belly pain, dark coloured urine, jaundice, and weight loss
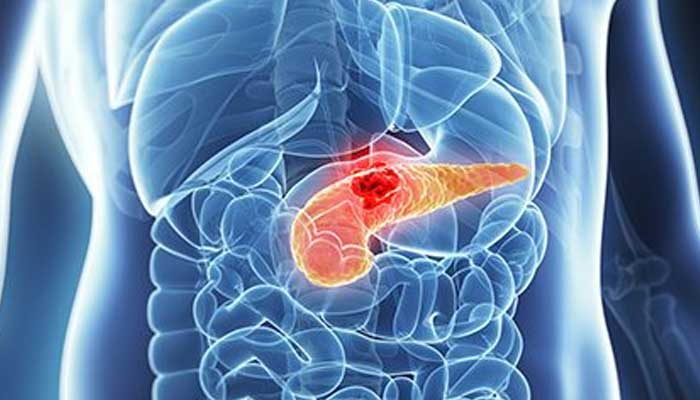
In a scientific breakthrough, a new medicine has been approved by the United States Foods and Drug administration (FDA) that can treat types of pancreatic cancer, Fox News reported.
Onivyde (irinotecan liposome) is the first medicine in nearly a decade that can treat the aggressive illness that has an average life expectancy of only eight to 11 months.
The drug was approved after a controlled trial was conducted on 770 patients with metastatic pancreatic adenocarcinoma and who had not previously received any chemotherapy.
The patients were injected with the new regimen which includes Onivyde, oxaliplatin, fluorouracil and leucovorin.
According to the results, the patients showed "significant improvements in survival rates and response rates".
Dr Zev Wainberg, professor of medicine and co-director of the UCLA GI Oncology Program in Los Angeles, said, "I am hopeful that this regimen represents a new reference, meaning we will add to this in the future."
However, Dr Marc Siegel, clinical professor of medicine at NYU Langone Medical Center, said that although "the drug is a useful new tool, it is not a game-changer."
Pancreatic cancer is the eighth most common type of cancer in females and tenth most common in males.
Its symptoms include loss of appetite, belly pain, dark coloured urine, jaundice, and weight loss.
-
Hantavirus outbreak: Could it be new pandemic threat like COVID? Experts weigh in
-
Meningitis leaves one dead, others critically ill; Know how it spreads
-
PCOS renamed PMOS: What new diagnosis means for millions of women’s health
-
Endometriosis linked to small increase in birth defect risk in Canadian study
-
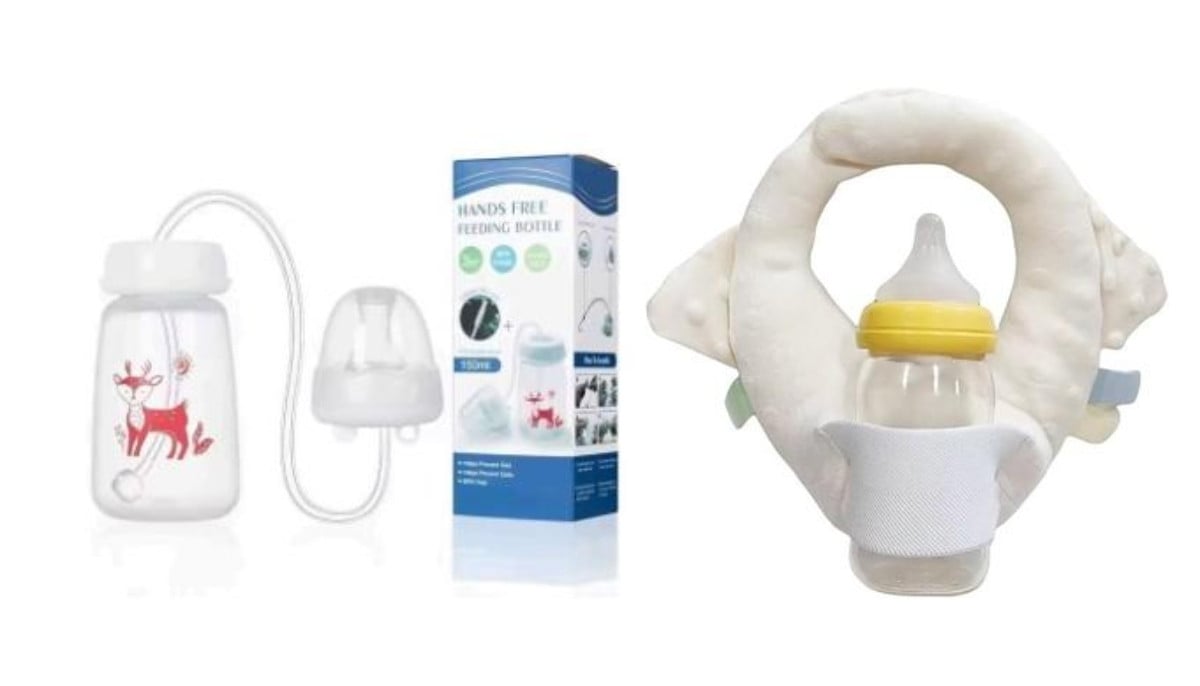
Health Canada issues safety warning over baby self-feeding products sold on Amazon
-
Do psychopaths and others have same brain structure? Scientists reveal shocking details
-
Cruise ship Hantavirus outbreak leaves 3 dead, more infected
-
43 hair products tested: Researchers find cancer-linked chemicals in braiding hair