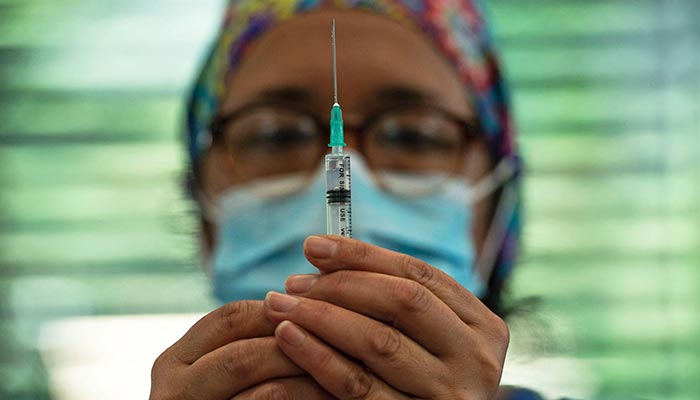
UK approves Pfizer bivalent Covid vaccine ahead of booster drive
The UK regulator last month okayed a similar bivalent vaccine by Moderna, becoming the first country to do so
LONDON: The UK's drug regulator said Saturday it had approved an updated Covid-19 jab by Pfizer/BioNTEch targeting the original virus and a subvariant ahead of an autumn booster drive.
The so-called "bivalent" jabs work for both the original virus that emerged in the Chinese city of Wuhan in 2019 and the BA.1 subvariant of Omicron.
The UK Medicines and Healthcare products Regulatory Agency (MHRA) approved the Pfizer jab as a booster for those aged 12 and over after a "careful review of the evidence".
The UK regulator last month okayed a similar bivalent vaccine by Moderna, becoming the first country to do so.
MHRA chief executive June Raine said: "I am pleased to announce that we now have a second approved vaccine for the UK Autumn booster programme."
The National Health Service in England and Scotland will begin a fresh booster rollout on Monday, starting with care home residents and eventually inviting everyone over 50.
The other UK regions of Wales and Northern Ireland are also launching similar campaigns.
The BBC reported Saturday that the Moderna bivalent shot would be offered first, but added that the UK did not have enough supplies to cover everyone over 50.
The European Union and the United States have recently authorised the updated version of the Pfizer vaccine and Moderna vaccines.
-
Meningitis leaves one dead, others critically ill; Know how it spreads
-
PCOS renamed PMOS: What new diagnosis means for millions of women’s health
-
Endometriosis linked to small increase in birth defect risk in Canadian study
-
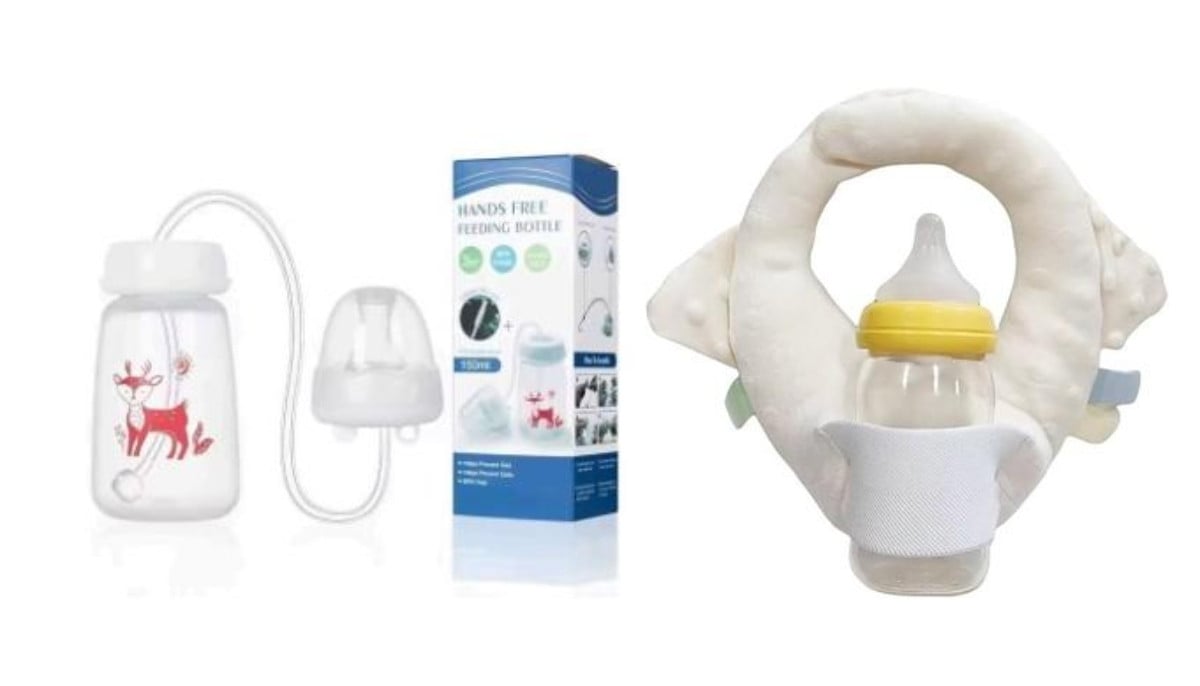
Health Canada issues safety warning over baby self-feeding products sold on Amazon
-
Do psychopaths and others have same brain structure? Scientists reveal shocking details
-
Cruise ship Hantavirus outbreak leaves 3 dead, more infected
-
43 hair products tested: Researchers find cancer-linked chemicals in braiding hair
-
Study reveals how brain tells you to stop scratching
-
Hantavirus outbreak: MV Hondius reaches Tenerife for ‘unprecedented’ evacuation
-
Omega-3 supplements linked to faster cognitive decline in high-risk patients
-
Is the US at risk? Everything you need to know about the new Hantavirus quarantines
-
Breakthrough research suggests, gum disease can be prevented without killing good bacteria