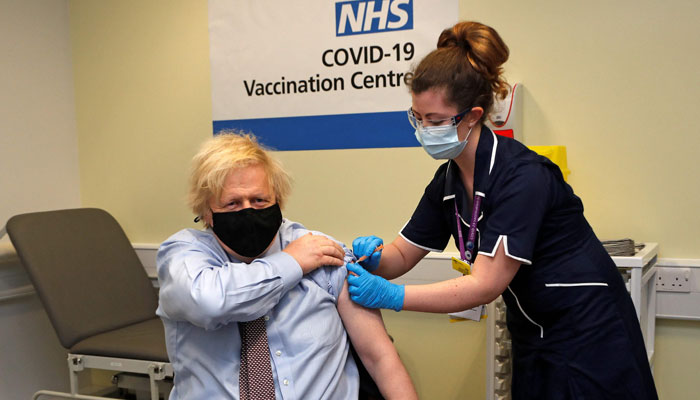
UK PM receives first dose of AstraZeneca vaccine
"The risk is Covid. This is a great thing to do," Boris Johnson says
LONDON: British Prime Minister Boris Johnson on Friday received a first dose of AstraZeneca's Covid vaccine, at the London hospital where he fought for his life almost a year ago, assuring the public the jab is safe.
"I literally did not feel a thing. It was very good, very quick," the 56-year-old leader told reporters at St Thomas´ Hospital in central London, close to his Downing Street residence.
"Everybody, when you do get your notification to go for a jab, please go and get it," Johnson said.
"It's the best thing for you, the best thing for your family and for everybody else," he added, stressing the renewed green light given by scientists in Europe for the AstraZeneca jab.
"The risk is Covid. This is a great thing to do," Johnson said, hours after French Prime Minister Jean Castex received the AstraZeneca injection live on television.
Johnson spent a week at St Thomas' Hospital, including three days in intensive care, after he was diagnosed with the coronavirus at the end of March last year.
He said after coming out that his personal battle with the coronavirus "could have gone either way", and there was "no question" doctors saved his life.
AstraZeneca's jab, developed with Oxford University, provides the bulk of Britain´s inoculation campaign and a large portion of its supply has been produced by the Serum Institute of India, the world´s biggest vaccine maker.
The institute this week said it could not provide planned deliveries next month, denting Britain's otherwise impressive campaign, which has delivered more than 26 million first doses.
The supply issues are another headache for AstraZeneca after its jab was suspended in several EU countries, pending a review by the European Medicines Agency following isolated cases of blood clots and brain haemorrhages.
The EMA restated its approval for the vaccine on Thursday, as did Britain's own drugs regulator.
-
Epstein victim Roza left traumatized by DOJ 'mistake'
-
Wes Streeting loses confidence in Starmer, resigns as UK Health Secretary
-
Iran opens Strait of Hormuz access to Chinese-linked tankers
-
US Treasury chief says Iranian ports blockade is working in putting pressure on Iran's economy
-
Iran urges BRICS to challenge West’s ‘false sense of superiority’
-
How China allowed sanctioned Marco Rubio into Beijing with a Chinese name for Trump visit
-
Israel-Lebanon talks kick off in Washington with US officials involved
-
White House says Trump and Xi aligned on Iran's nuclear threat